These rare but interesting lesions comprise 1 per cent of all brain 'tumors'. The spectrum of developmental cysts in CNS include, arachnoid cysts, ependymal cysts, colloid cysts, dermoid and epidermoid cysts, Dandy walker cysts ,epithelial cysts, and porencephalic cysts. They are mostly intracranial, but can occur intraspinally as well.
1) ARACHNOID CYSTS (leptomeningeal cysts):
They were referred by Richard Bright in 1831 as ‘serous cysts of the arachnoid?? he described cystic lesions with liquid, clear, contents and intra-arachnoidal localization. About 127 years later, Starkman described the intra-arachnoid localization of these cysts, giving an explanation for their etiology.
Epidemiology:
Arachnoid cysts correspond to about 1% of all intracranial lesions in the general population and about 3% in the pediatric population. Although 60-90% of reported cases have been found in young people (less than 20 yeas old), it is possible to find them in the elderly.
The sex distribution shows a preference for the males, in the ratio of 3:1 and for left side of the CNS structures.
Etiology:
They are congenital lesions that probably arise during development from splitting or duplication of this membrane.
The etiology of these lesions is extremely controversial.
Intra-arachnoid cyst theory theory suggests a congenital origin for the etiology of the arachnoid cysts; at some time during development, there is a duplication of the arachnoid membrane, proliferation of the arachnoid cells, forming a cavity which later fills with CSF through several mechanisms. This was defended by Starkman and suggested by Bright in 1831. Frequently these cysts are associated with venous anomalies and the developmental anomaly probably occurs between the sixth and eighth week of fetal life, precisely when the vascular structures begin their development.
Shreiber, suggested that most arachnoid cysts were caused by incomplete canalization of the subarachnoid space with the formation or pouch like spaces which become distended with CSF consequent upon arterial pulsation.
Dott and Gillingham, also postulated that such pouches usually form along the axis of a main cerebral artery, that the leptomeningeal space at the distal boundary of a cistern becomes occluded and that the fluid propelled into the proximal portion cannot escape over the convexity of the brain.
The striking and nearly invariable association of arachnoid cysts with normal subarachnoid cisterns has led to hypothesize that arachnoid cysts represent a congenital anomaly of the developing subarachnoid cisterns in early intrauterine life. It is postulated that, during the process of the complex folding of the primitive neural tube and the formation of normal subarachnoid cisterns, an anomalous splitting of the arachnoid membrane occurs.
Cysts of the sellar region (supra, para and intrasellar) are considered to be an extension of the Lillequist membrane, which divides the chiasmatic cistern of the interpeduncular cistern, being associated with alterations of the normal CSF flow.
Cysts of the posterior fossa are probably caused by various degrees of obstruction of the foramens of Luschka or Magendie. Intraventricular cysts are explained by mesenchymatous invagination through the choroidal tissue that is formed by digitations of pia-mater with arachnoidal stroma.
Spinal intradural cysts are related to the septum posticum at a thoracic level, and thoracolumbar extradural cysts are explained as invaginations of the arachnoid through small pre-existent dural defects.
The fact that the arachnoid cysts occur mainly in young people (less than 20 years old) with a strong incidence in the first years of life as well as the occurrence of case reports with a familiar incidence and the frequent association of these cysts with diseases related to chromosome alterations (polycystic kidney, neurofibromatosis, chromosome 12 trisomy) reinforce the possibility of a congenital origin for these lesions.
Apart from the structural alterations there is an alteration of the normal CSF flow, as confirmed in some cases, by the persistence of symptoms in patients whose cysts were surgically removed.
Pathology:
|
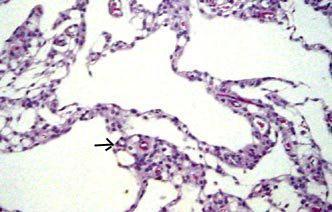
An arachnoid cyst is by definition, a benign lesion, with well defined outlines, within the arachnoid membrane or covered by layers of arachnoid cells supported by collagen fibres, having liquid contents similar to CSF. It is a cavity, whose walls are formed by arachnoid cells (simple or multiple layers), supported by a stroma, rich in collagen fibres.
They can develop anywhere along the cerebro-spinal axis, but almost all occur in relation to an arachnoid cistern.
The distribution of arachnoid cysts in two hundred and eight reported cases is as follows:
Sylvian fissure, 49%; cerebellopontine angle, 11%; supracollicular area, 10%; the vermis, 9%; sellar and suprasellar area, 9%; interhemispheric fissure, 5%; cerebral convexity, 4%; and the clival and interpeduncular area, 3%.
|
|
|
|
Lobulated arch.cyst(H&E)- lining of meningothelial (arrow)
cells supported by collagen fibres.
|
|
A lesser percentage occurs within the ventricular system. Intrasellar cysts are the only intracranial arachnoid cysts that are extradural. Intraspinal cysts are rarer.
The structural features of the arachnoid cyst wall that distinguish it from the normal arachnoid membrane are
(1) splitting of the arachnoid membrane at the margin of the cyst,
(2) a very thick layer of collagen in the cyst wall,
(3) the absence of traversing trabecular processes within the cyst, and
(4) the presence of hyperplastic arachnoid cells in the cyst wall, which presumably participate in collagen synthesis.
Most of the small arachnoid cysts do not increase in size and can be asymptomatic, but the larger cysts can become bigger and produce symptoms.
The possible reasons for the increase in size:
The active secretion through the cyst wall inside the cavity
Unidirectional valve mechanism (ball valve) with CSF flow from the sub-arachnoid space into the cyst
The active osmosis through the wall by a similar mechanism to that which occurs at the Pacchionian granulations
Pulsations of the CSF and of the intracranial arteries transmitted to the cyst cavity through a wide opening into the sub-arachnoid space.
Transudation through the wall cyst from the choroids plexus (in the intraventricular cysts).
It is not known which mechanism is responsible for the increase of the cyst volume, which is probably multifactorial. However in intraventricular cysts related to the choroid plexus, the principal mechanism is the transudation through the cyst wall from plexus secretion. For cysts that are in communication with the sub-arachnoid space, the principal mechanism is likely to be the ‘bell valve??effect and in non-communicating cysts, active secretion mechanism or osmosis may be more likely.
A classification based on cisternography with metrizamide and CT isotopic cisternography and, more recently cine MRI, defines the existence of communication with the subarachnoid space and the cavity of the cyst. The cyst can then be as slowly or rapidly communicating or non communicating (real cysts).
A classification based on etiology divides the cysts into primary (congenital) or secondary (traumatic or infectious origin).
Finally a classification which is based on the morphology, volume and effect on the CNS structures and bone structure as suggested by Galassi divides the cysts into 3 types. Type 1 are small asymptomatic cysts, type II have some mass effect, with bone erosion and type III have deformation of large areas of the CNS and gross bony changes.
Clinical features:
In many instances, an arachnoid cyst is an incidental finding. The clinical presentation will depend on the location and size of the
achnoid cyst, and the symptoms often are mild considering the large size of some cysts. Most patients will come to medical attention in the first two decades of life, often in the first 6 months.
These lesions cause symptoms and signs of increased ICP by compressing the normal tissue and obstructing the CSF pathway.
The symptoms depend on the cyst localization. Symptoms and signs include cranial enlargement, localized cranial bulging, especially the large cysts, can present acutely with sudden deterioration.
Suprasellar cysts may also present with endocrine symptoms, head bobbing, and visual disturbances. Either communicating or obstructive hydrocephalus is often present. In the elderly, dementia has been described.
Intraspinal cysts may produce a tetra or para paresis, with abnormal reflexes, sphincter dysfunction, sensibility alterations and radicular pain according to the level of the lesion.
Investigations:
Arachnoid cysts are usually diagnosed by CT or MRI. Further evaluation with CSF contrast flow studies is only occasionally necessary for the diagnosis of midline suprasellar and posterior fossa lesions.
CT shows a smoothly bordered noncalcified extraparenchymal cystic mass with density similar to CSF and no contrast enhancement and also the bony remodeling.
MRI may show the arachnoid membrane and also differentiates the CSF in the arachnoid cysts from the neoplastic cysts and ependymal ( usually, intraparenchymal) cysts. Porencephalic cyst usually communicate into the ventricle. In addition associated cerebral and cerebellar hypoplasias are well studied.
Deep invagination of an arachnoid cyst into the cerebral hemisphere may simulate porencephaly to such an extent that it has been termed pseudoporencephaly. However, the inferior aspect of the arachnoid cyst shows a displaced but otherwise normal cerebral cortex, while in porencephaly, the surrounding cortex and white matter are abnormal.
Management:
The cysts, that do not cause mass effect and have not changed in size need not be treated.
Many procedures for treatment of arachnoid cysts have been proposed. Surgical options for arachnoid cysts include drainage by needle aspiration, craniotomv with excision of the wall and fenestration into the basal cisterns (open or endoscopically) with or without a silastic shunt, and shunting of the cyst into the peritoneum or vascular system.
The Fenestration and shunting are the two most common options.
Shunting of the cyst material into the peritoneum or into the vascular system is associated with low morbidity and mortality and a relatively low rate of recurrence, but the patient becomes shunt dependant. Long-term complications of a shunt occur in more than one third of cases. Low pressure shunt is preferred.
Craniotomy with excision of the cyst wall and fenestration into the basal cisterns permits direct inspection of the cyst and avoids placement of a permanent shunt in some cases. However, it is associated with reaccumulation of CSF at the cyst site. In addition, significant morbidity and mortality may accompany; abrupt displacement of brain structures following the rapid decompression that accounts for the unexpected rapid deterioration.
Endoscopic approaches have been used with good results, although the follow-up period has not been long. The goal of fenestration is free communication with the basal cisterns.
Shunting the associated hydrocephalus alone will only worsen the symptoms, and may increase the size of the arachnoid cyst.
Outcome:
Arachnoid cysts are benign lesions, with a poorly defined natural history, sometimes with spontaneous disappearance.
Regression of cyst volume post-surgery, occurs independently of the surgical technique. Sometimes the cyst volume remains constant, leaving the patient without symptoms. Usually there is a good clinical and imaging correlation of regression.
For the suprasellar cysts with hydrocephalus, it is common to have persistent ventricular dilatation in spite of the cyst having decreased in its volume, with normal intracranial pressure. This has no consequences in the intellectual development of the patients. For the sellar cysts it is also common to have persistence of endocrinological alterations, even with cyst disappearance.
Arachnoid Cysts by location:
Sylvian fissure/Middle cranial fossa cysts:
The sylvian fissure is the most common site for arachnoid cysts (50% of adult cases and 30% of pediatric cases). They are usually small or medium, but they can become quite large and open up the fissure to expose the insula and middle cerebral branches. They usually manifest clinically in children or adolescents, but can present at any age. Males predominate and the left hemisphere is more commonly involved.
Global headache, seldom severe, is common. Macrocephaly or an asymmetric macrocrania is often the presentation in the young.
Children may present with developmental delay. Acute symptoms of increased ICP, hemiparesis, or seizures are common.
On CT and MRI, the large cysts extend posteriorly and open up the sylvian fissure. The temporal lobe may be underdeveloped. The greater wing of the sphenoid may be displaced anteriorly and the lesser wing may be elevated. In most cases, the associated hydrocephalus is due to compression of the 3rd ventricle.
Treatment is either shunting the cyst or fenestration of the cyst into a cistern or both combined. Symptoms typically improve after successful treatment. Results are poor when behavioral abnormalities and mental retardation are present.
Suprasellar cysts:
Suprasellar location accounts for approximately 10% of arachnoid cysts and less than 1% of intracranial mass lesions. Cysts in the suprasellar region appear to arise from the suprasellar cistern. It has been suggested that the cysts may arise from an imperforate membrane of Lillequist.
Typically, most patients are adolescents, and present with hydrocephalus. Adults present with visual problems. Hypopituitarism, especially the growth hormone deficiency, can occur.
Rarely, there may be ‘bobble head-doll?? syndrome. They experience an involuntary two or three times per second vertical bobbing of the head, with compensatory horizontal movements of the trunk, attributed to the abnormal pressure exerted by the cyst on the 3rd ventricle and on the dorsomedial nucleus of the thalamus.
On CT and MRI images, a suprasellar arachnoid cyst appears as a smooth, oval or round lesion in the region of the third ventricle .
Multiple approaches to suprasellar cyst fenestration have been attempted, including transfrontal removal of the anterior portion of the cyst, a transcallosal approach to communicate the cavity of the cyst into the ventricular system, and insertion of catheters between the cyst and ventricle or chiasmatic cisterns. Any one of these operations has a poor chance of prolonged success and may fail to decrease either the size of the cyst or the ventricular system.
Shunting the associated hydrocephalus will only worsen the symptoms, and may increase the size of the arachnoid cyst. A combination of cyst fenestration and ventricular shunting may be more successful.
Convexity and Interhemispheric fissure cysts:
Convexity lesions differ from arachnoid cysts in other locations due to their lack of contact with cisternal spaces. Determining the origin of interhmispheric cysts can be difficult. Imaging of the corpus callosum, third ventricle, and collicular plate is essential, because the third ventricle can herniate into the interhemispheric fissure, mimicking an arachnoid cyst.
Usually, they are incidental findings. Adults can present with raised ICP, epilepsy, and focal neurological deficit. Children may present with localized macrocrania.
CTand MRI demonstrate rounded lesions with CSF density, overlying the cerebral cortex or interhemisheric area.
Typically, they are treated with cysto-peritoneal shunting, often with excision of the outer cyst wall.
Most of these cysts have been detected in patients aged less than 15 years. Symptoms frequently arise from hydrocephalus secondary to compression of the posterior third ventricle or the aqueduct.
Quadrigeminal plate arachnoid cysts:
Pupillary reactivity or eye movements are disturbed due to compression of the quadrigeminal plate or stretching of the 4th nerve.
|
CT and MRI show an ovoid collection posterior to the 3rd ventricle.
Definitive surgical treatment can be difficult because the region is not easily accessible. Fenestration followed by shunting may have a better success. Recurrence is high.
Intraventricular arachnoid cysts:
The differential diagnosis includes ependymal cyst, epidermoid cyst, dermoid cyst, infectious cyst, and porencephalic cyst.
MRI is the definitive study. Asymptomatic cavum septi pellucidi, and septum cavum veli
interpositi may be considered normal variants.
|
|
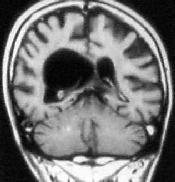
|
|
Intraventricular arach.cyst
|
|
Simple drainage is usually followed by reaccumulation. Endoscopic fenestration, craniotomy and cyst excision, cyst-peritoneal shunting are the surgical options.
Posterior fossa archnoid cysts:
They are less common, about 25% of all intracranial cysts and usually occur in the midline, superficial to the vermis or in the CP angle. Less frequent locations include the cerebellar convexities, or pre pontine areas. The posterior fossa is a common site of benign intracranial cysts, especially in children.
The differential diagnosis of a posterior fossa include, arachnoid cysts, Dandy-Walker malformation, and mega cisterna magna.
|
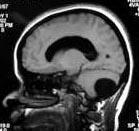
|
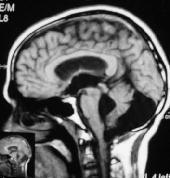
|
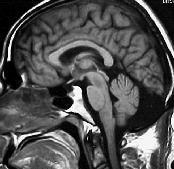
|
|
Post. fossa arch. cyst- MRI
|
Dandy-walker cyst-MRI
|
Mega cisterna magna-MRI
|
|
An arachnoid cyst results in anterior displacement of the fourth ventricle, but normal cerebellar development.
|
Dandy-Walker malformation is a cystic dilatation of the fourth ventricle or a cyst in communication with 4th ventricle.
|
Mega cisterna magna is an anatomic variant with normal fourth ventricle and small cerebellum.
|
Most pediatric patients present with macro crania. In adults, there are intermittent symptoms or influenced by posture. CP angle lesions can cause tinnitus or hearing loss.
Fenestration, cyst shunting, and combination of shunting and fenestration are the treatment options. A ventricular shunting is often necessary, as the hydrocephalus often fails to resolve with cyst decompression. Deeper cysts are often multiloculated, and probably would not respond to shunting and are usually treated with fenestration.
Spinal arachnoid cysts:
|
Arachnoid cysts also occur within the spinal canal, in which arachnoid cysts or arachnoid diverticula may be located subdurally or in the epidural space, respectively. They are rarer than intracranial cysts. Extramedullary location is common. They are mostly congenital in origin.
Typically, spinal arachnoid cysts occur at the midthoracic level and, less frequently, at the lumbosacral or sacral level. Commonly located dorsal to the cord. A cyst in this location is usually secondary to a congenital or acquired defect and is situated in an extradural location. Intradural spinal arachnoid cysts are secondary to a congenital deficiency within the arachnoid or are the result of adhesions resulting from previous infection or trauma.
|
|
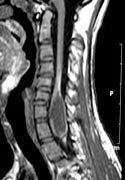
|
|
Intramedullary Spinal Archnoid cyst
|
|
They cause symptoms indistinguishable from cord compression due to other causes. Patients with spinal arachnoid cysts may become symptomatic as a result of local cord displacement or cord compression. Epidural arachnoid cysts often are associated with kyphoscoliosis in juveniles. Arachnoid cysts also are associated with myelodysplasia in spinal dysraphic lesions. Pain produced by intraspinal arachnoid cysts typically is aggravated by the Valsalva maneuver, which increases pressure within the cyst.
Remission of symptoms is not uncommon. Surgical excision is curative.
Tarlov cysts (Spinal perineurial cyst):
Many consider this a degenerative cyst and not developmental.
Tarlov described this cyst in 1938, while conducting an anatomic study of the filum terminale. It is a cystic dilatation of the subarachnoid space around a nerve root (between the perineurium of the nerve root and the outer surface of its pia). The cyst can dissect into the nerve and can contain nerve fibers within it. These cysts typically involve the sacral nerve roots. It is found on as many as 5% of lumbosacral MRI.
It is usually diagnosed incidentally. Approximately, 4 out of 5, are asymptomatic. Some may present with radicular pain, frequently occurring in attacks with pain free intervals. Many patients get relief while by assuming Trendelenberg position in which the patient is on an elevated and inclined plane, usually about 45°, with the head down and legs and feet over the edge of the table. Incontinence.& pain on moving the sacrum are seen in some patients.
Although a spinal perineurial cyst involves a single nerve root at first, it may enlarge to the point where it compress adjacent nerve roots as well.
It is difficult to prove that Tarlov cysts cause symptoms in many cases because other findings that can cause these symptoms (disc herniation, stenosis.) are usually found along with the cyst on MR images or at surgery.
Surgery indicated only if they cause progressive or disabling symptoms.
Percutaneous CT-guided drainage provides good relief for several months in most patients but cysts recurs in almost all. Although the cysts re-pressurize and the patients' symptoms returned in most cases, this technique seems to be a quick and simple way of at least attaining a pain-free interval and possibly a complete cure in some patients.
Sacral laminectomy with microsurgical cyst fenestration and closure with reinforced epidural fat or muscle grafts and fibrin glue application is widely employed; lumbar drains for cerebrospinal fluid diversion for several days postoperatively has also been suggested.
Secondary (False) arachnoid cysts:
Arachnoid cysts that are not congenital have been termed secondary or false cysts. I feel a brief mention of these acquired cysts is appropriate in this section of developmental cysts.
Such cysts represent accumulations of CSF resulting from postinflammatory loculation of the subarachnoid space in patients with head injury, infection, or ICH. The cyst membrane may also be composed of arachnoid, but in addition, inflammatory cells and hemosiderin deposits may be present and it is difficult to fenestrate these cysts.
Lepto meningeal cysts associated with growing fractures in children are rare. The physiologic brain growth and CSF pulsations contribute to cyst herniations through a dural rent. Treatment includes primary repair of the dural rent and the bony defect.
2) EPENDYMAL CYSTS:
|
These rare cysts arise due to the inclusion of ependymal cells in the substance of the brain. They may also originate from ectopic glial tissue present in the subarachnoid space. Ependymal cysts are common in oral-facial-digital syndromes.
These later take up a secretory function resulting in the formation of a cyst. These are intracerebral and usually adjacent to the ventricles with which they may occasionally communicate.
The gross appearance is usually indistinguishable from an arachnoid cyst. Microscopically, the cyst may be lined by cells resembling ependyma in some places. They may bear cilia. Ultrastructural studies confirm the neuroepithelial origin of these cysts.
|
|
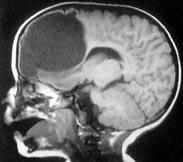
|
|
Frontal ependymal cyst
|
|
Colloid cysts of the third ventricle have also been termed neuroepithelial cysts. Cysts in the fourth ventricle which were lined by cells similar to ependymal cells and cuboidal cells resembling the epithelium of the choroid plexus have been reported. Similar choroid plexus cysts may also occur in the third ventricle and block the foramen of Munro.
Excision is required in only the symptomatic cases. The rest may be followed up periodically.
3) RATHKE'S CLEFT CYSTS:
These cysts were first described by Martin Heinrich Rathke (1793??860), a German anatomist. Rathke's cleft cysts are benign, nonneoplastic lesions that are believed to be remnants of Rathke's pouch, which is the superiorly directed evagination from the stomadium of the 4 week old human embryo; all but the cranial portion of the pouch becomes obliterated by week 7 of gestation. The anterior wall of the remaining cavity becomes the anterior pituitary, while the posterior wall becomes the pars intermedia of the gland.
Rathke’s cleft cysts are primarily intrasellar and are found in 13% to 23% of postmortem examinations.
Rathke's cleft cysts are uniloculate and thin-walled and contain watery to mucinous fluid. Light microscopy of the lining shows goblet, ciliated and, to a lesser extent, secretory cells of anterior pituitary type. The finding of ciliated epithelial and mucous secreting cells in a pituitary gland are pathgnomonic for Rathke's cleft cysts.
Radiologically, they may mimic craniopharyngiomas or as cystic pituitary adenoma.
Plain skull radiographs do not, usually, reveal a enlarged sella turcica. In patients with symptomatic Rathke's cleft cysts , plain skull radiographs commonly demonstrate findings of an abnormally configured sella, which varies from slight asymmetry of the sellar floor to massive erosion. In some patients, intrasellar and/or suprasellar calcification is observed.
CT reveals an hypodense cystic mass lesions arising from the pituitary fossa, without enhancement.
MRI shows cystic lesions with long T1 and long T2 although sometimes intrinsic paramagnetic substances may produce T1 and T2 shortening resulting in hyperintense T1 and hypointense T2 lesions. Rathke's cleft cysts usually have a thin wall that may enhance with gadolinium-based contrast material. Variability in the gadolinium enhancement among individual cysts may reflect squamous metaplasia in the wall or a peripherally displaced rim of pituitary tissue.
Rathke's cleft cysts almost always are homogeneous in signal intensity, whereas other lesions, such as cystic craniopharyngiomas and hemorrhagic adenomas, more frequently have heterogeneous signal intensity.
These cysts are rarely symptomatic and are usually incidental findings on imaging studies. Occasionally, they cause symptoms by causing pressure on adjacent structures such as the optic nerves and the pituitary gland causing visual problems, loss of pituitary function, hypothalamic dysfunction and headache when they grow in the suprasellar space. A Rathke's cyst may occasionally be associated with a pituitary adenoma. Partial excision of the cyst wall and drainage should result in cure: only a small percentage of such cysts recur.
4) PORENCEPHALIC CYSTS:
|
More appropriate term may be 'hole in the brain'
Porencephalic cysts, characteristically, communicate with the ventricles or subarachnoid space and are covered on the outside by arachnoid.
These are congenital (primary) intracranial cysts and may arise as a leptomeningeal cyst.
It is possible that a failure of development of a part of the cerebral mantle may result in a cyst.
In addition to their congenital origin, porencephalic cysts may also arise as a result of trauma specially during birth or infancy when following loss of cerebral tissue adjacent to the ventricles a cyst forms and communicates with the ipsilateral dilated ventricle.
Puncture porencephaly is the development of a cystic cavitation along the track of a ventricular needle, manifesting in course of time, following rise in intracranial pressure due to nonfunctioning of the shunt in cases of hydrocephalus.
|
|
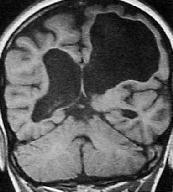
|
|
Porencephalic cyst
|
|
This may also follow prolonged ventricular drainage or repeated ventricular punctures.
Surgical excision may be considered in cases where these cysts are found to be the cause of intractable epilepsy. The area of excision should include the surrounding gliosed cerebral tissue as well.
5) Other developmental cysts are discussed elsewhere.
They are, Colloid cyst of the third ventricle, Dandy Walker cyst, and Epidermoids, Dermoids, & Neuroenteric cysts.